Cu-Mg Example¶
The Cu-Mg binary system is an interesting and simple binary subsystem for light metal alloys. It has been modeled in the literature by Coughanowr et al. [1], Zuo and Chang [2] and Zhou et al. [3] and was featured as a case study in Computational Thermodynamics The Calphad Method by Lukas, Fries, & Sundman [4].
Here we will combine density functional theory and experimental calculations of single-phase data to generate a first-principles phase diagram. Then that database will be used as a starting point for a Markov Chain Monte Carlo (MCMC) Bayesian optimization of the parameters to fit zero-phase fraction data.
Input data¶
All of the input data for ESPEI is stored in a public ESPEI-datasets repository on GitHub. The data in this repository is Creative Commons Attribution 4.0 (CC-BY-4.0) licensed and may be used, commercialized or reused freely.
In order to run ESPEI with the data in ESPEI-datasets, you should clone this repository to your computer. Files referred to throughout this tutorial are found in the CU-MG folder. The input files will be very breifly explained in this tutorial so that you are able to know their use. A more detailed description of the files is found on the Gathering input data page.
If you make changes or additions, you are encouraged to share these back to the ESPEI-datasets repository so that others may benefit from this data as you have. You may then add your name to the CONTRIBUTORS file as described in the README.
Phases and CALPHAD models¶
The Cu-Mg system contains five stable phases: Liquid, disordered fcc and hcp, the C15 Laves phase and the CuMg2 phase. All of these phases will be modeled as solution phases, except for CuMg2, which will be represented as a stoichiometric compound. The phase names and corresponding sublattice models are as follows:
LIQUID: (CU, MG)1
FCC_A1: (CU, MG)1 (VA)1
HCP_A3: (CU, MG)1 (VA)1
LAVES_C15: (CU, MG)2 (CU, MG)1
CUMG2: (CU)1 (MG)2
These phase names and sublattice models are described in the JSON file Cu-Mg-input.json file as seen below
{
"components": ["CU", "MG", "VA"],
"phases": {
"LIQUID" : {
"sublattice_model": [["CU", "MG"]],
"sublattice_site_ratios": [1]
},
"CUMG2": {
"sublattice_model": [["CU"], ["MG"]],
"sublattice_site_ratios": [1, 2]
},
"FCC_A1": {
"sublattice_model": [["CU", "MG"], ["VA"]],
"sublattice_site_ratios": [1, 1]
},
"HCP_A3": {
"sublattice_site_ratios": [1, 0.5],
"sublattice_model": [["CU", "MG"], ["VA"]]
},
"LAVES_C15": {
"sublattice_site_ratios": [2, 1],
"sublattice_model": [["CU", "MG"], ["CU", "MG"]]
}
}
}
ESPEI¶
ESPEI has two types of fitting – parameter generation and MCMC optimization. The parameter generation step uses experimental and first-principles data of the derivatives of the Gibbs free energy to parameterize the Gibbs energies of each individual phase. The MCMC optimization step fits the generated parameters to experimental phase equilibria data. These two fitting procedures can be used together to fully assess a given system. For clarity, we will preform these steps separately to fit Cu-Mg. The next two sections are devoted to describing ESPEI’s parameter generation and optimization.
Though it should be no problem for this case, since the data has already been
used, you should get in the habit of checking datasets before running ESPEI.
ESPEI has a tool to help find and report problems in your datasets. This is
automatically run when you load the datasets, but will fail on the first error.
Running the following commmand (assuming from here on that you are in the
CU-MG folder from ESPEI-datasets):
espei --check-datasets input-data
The benefit of the this approach is that all of the datasets will be checked and
reported at once. If there are any failures, a list of them will be reported
with the two main types of errors being JSONError, for which you should read
the JSON section of Gathering input data, or DatasetError, which are related to
the validity of your datasets scientifically (maching conditions and values
shape, etc.). The DatasetError messages are designed to be clear, so please
open an issue on GitHub
if there is any confusion.
First-principles phase diagram¶
By using the Cu-Mg-input.json phase description for the fit settings and
passing all of the input data in the input-data folder, we can first use
ESPEI to generate a phase diagram based on single-phase experimental and DFT
data. Currently all of the input datasets must be formation properties, and it
can be seen that the formation enthalpies are defined from DFT and experiments
for the Laves and CuMg2 phases. Mixing enthalpies are defined for the for the
fcc, hcp, and Laves phases from DFT and for liquid from experimental
measurements.
The following command will generate a database named cu-mg_dft.tdb with
parameters selected and fit by ESPEI:
espei --input espei-in.yaml
where espei-in.yaml is a ESPEI input file with
the following contents
system:
phase_models: Cu-Mg-input.json
datasets: input-data
generate_parameters:
excess_model: linear
ref_state: SGTE91
output:
output_db: cu-mg_dft.tdb
The calculation should be relatively quick, on the order of a minute of runtime.
With the above command, only mininmal output (warnings) will be reported. You
can increase the verbosity to report info messages by setting the
output.verbosity key to 1 or debug messages with 2.
With the following code, we can look at the generated phase diagram and compare it to our data.
# First-principles phase diagram
from pycalphad import Database, variables as v
from espei.datasets import load_datasets, recursive_glob
from espei.plot import multiplot
import matplotlib.pyplot as plt
# load the experimental and DFT datasets
datasets = load_datasets(recursive_glob('input-data', '*.json'))
# set up the pycalphad phase diagram calculation
dbf = Database('cu-mg_dft.tdb')
comps = ['CU', 'MG', 'VA']
phases = ['LIQUID', 'FCC_A1', 'HCP_A3', 'CUMG2', 'LAVES_C15']
conds = {v.P: 101325, v.T: (300, 1500, 10), v.X('MG'): (0, 1, 0.01)}
# plot the phase diagram and data
multiplot(dbf, comps, phases, conds, datasets)
plt.savefig('cu-mg_dft_phase_diagram.png')
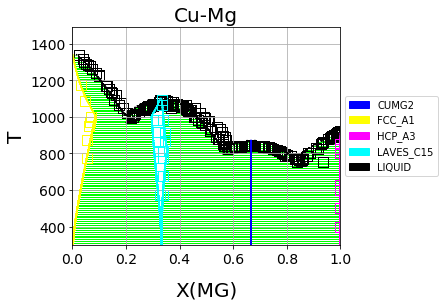
Which should result in the following figure
We can see that the phase diagram is already very reasonable compared to the experimental points. The liquidus temperatures and the solubilities of the fcc and Laves phases are the key differences between the equilibrium data and our first-principles phase diagram. The next section will discuss using ESPEI to optimize the parameters selected and calculated based on the single-phase data to these multi-phase equilibria.
MCMC optimization¶
With the data in the CU-MG input data, ESPEI generated 18 parameters to fit. For systems with more components, solution phases, and input data, may more parameters could be required to describe the thermodynamics of the specific system well. Because they describe Gibbs free energies, parameters in CALPHAD models are highly correlated in both single-phase descriptions and for describing equilibria between phases. For large systems, global numerical optimization of many parameters simultaneously is computationally intractable.
To combat the problem of optimizing many paramters, ESPEI uses MCMC, a stochastic optimzation method. For more details on how MCMC is used in ESPEI, see the Theory page.
Now we will use our zero phase fraction equilibria data to optimize our
first-principles database with MCMC. The following command will take the
database we created in the single-phase parameter selection and perform a MCMC
optimization, creating a cu-mg_mcmc.tdb:
espei --input espei-in.yaml
where espei-in.yaml is an ESPEI input file with
the following structure
system:
phase_models: Cu-Mg-input.json
datasets: input-data
mcmc:
mcmc_steps: 1000
input_db: cu-mg_dft.tdb
output:
output_db: cu-mg_mcmc.tdb
ESPEI defaults to run 1000 iterations and depends on calculating equilibrium in pycalphad several times for each iteration and the optimization is compute-bound. Fortunately, MCMC optimzations are embarrasingly parallel and ESPEI allows for parallelization using dask or with MPI using mpi4py (single-node only at the time of writing - we are working on it).
Note that you may also see messages about convergence failures or about droppping conditions. These refer to failures to calculate the log-probability or in the pycalphad solver’s equilibrium calculation. They are not detrimental to the optimization accuracy, but overall optimization may be slower because those parameter steps will never be accepted (they return a log-probability of \(-\infty\)).
Finally, we can use the newly optimized database to plot the phase diagram
# Optimized phase diagram from ESPEI's multi-phase fitting
from pycalphad import Database, variables as v
from espei.datasets import load_datasets, recursive_glob
from espei.plot import multiplot
import matplotlib.pyplot as plt
# load the experimental and DFT datasets
datasets = load_datasets(recursive_glob('input-data', '*.json'))
# set up the pycalphad phase diagram calculation
dbf = Database('cu-mg_mcmc.tdb')
comps = ['CU', 'MG', 'VA']
phases = ['LIQUID', 'FCC_A1', 'HCP_A3', 'CUMG2', 'LAVES_C15']
conds = {v.P: 101325, v.T: (300, 1500, 10), v.X('MG'): (0, 1, 0.01)}
# plot the phase diagram and data
multiplot(dbf, comps, phases, conds, datasets)
plt.savefig('cu-mg_mcmc_phase_diagram.png')
ESPEI allows thermodynamic database to be easily reoptimized with little user interaction, so more data can be added later and the database reoptimized at the cost of only computer time. In fact, the existing database from estimates can be used as a starting point, rather than one directly from first-principles, and the database can simply be modified to match any new data.
References¶
| [1] | Coughanowr, C. A., Ansara, I., Luoma, R., Hamalainen, M. & Lukas, H. L. Assessment of the Cu-Mg system. Zeitschrift f{ü}r Met. 82, 574–581 (1991). |
| [2] | Zuo, Y. U. E. & Chang, Y. A. Thermodynamic calculation of the Mg-Cu phase diagram. Zeitschrift f{ü}r Met. 84, 662–667 (1993). |
| [3] | Zhou, S. et al. Modeling of Thermodynamic Properties and Phase Equilibria for the Cu-Mg Binary System. J. Phase Equilibria Diffus. 28, 158–166 (2007). doi:10.1007/s11669-007-9022-0 |
| [4] | Lukas, H., Fries, S. G. & Sundman, B. Computational Thermodynamics The Calphad Method. (Cambridge University Press, 2007). doi:10.1017/CBO9780511804137 |
Acknowledgements¶
Credit for initially preparing the datasets goes to Aleksei Egorov.